
The MICT® technology system is a desktop based, in vitro diagnostic device. The purpose of the MICT® system, when used with its disposable Test Devices, is to provide a means to quantitatively and qualitatively detect biochemical analytes directly from body fluids or environmental samples.
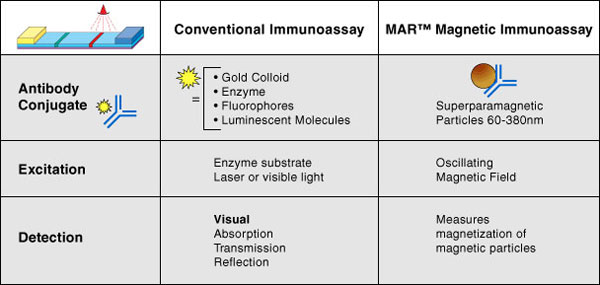
The format of MICT® assays is similar to that of standard home pregnancy tests. In MICT® assays, however, small magnetic nanoparticles replace the gold or colored latex particles used in visually read tests. These nanoparticles have similar flow and capture characteristics to their colored analogues, however in the MICT® technology system, a magnetic measurement of the capture line is made. The magnetic measurement is directly related (quantitatively) to the amount of accumulated magnetic material in the capture region.
The magnetic measurement requires the excitation of the nanoparticles in a magnetic field. This magnetic field is provided by way of a C-shaped electromagnet, while the magnetic measurement is performed by an array of thin film induction coils. The format of the capture line, as well as the geometry, position, material selection and electronics, all contribute to the sensitive nature and quantitative ability of the measurement.
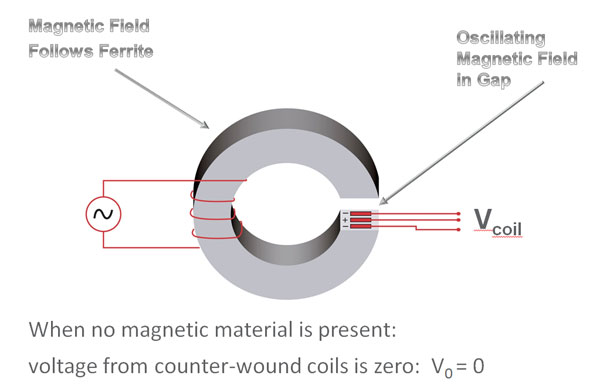
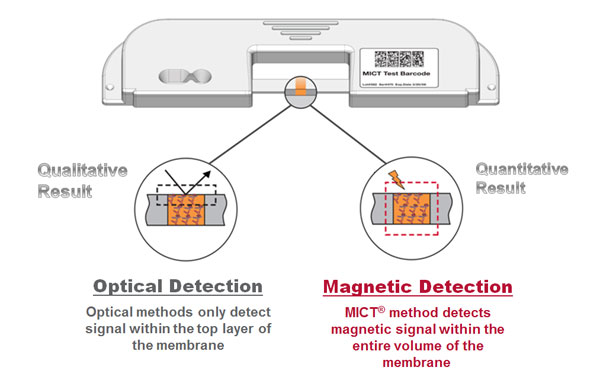
The graphic below helps to illustrate the sensitivity advantage offered by MICT®. In all immuno-chromatographic tests, the capture region extends throughout the depth of the membrane, as does particle accumulation in the completed test. In optically read assays (visual or reflectance), only the accumulation of particles on the top surface can be detected. Typically depth resolution is limited to 10μ in membranes that are approximately 200 μ thick. In MICT®, the entire particle distribution can be resolved by way of a magnetic measurement:
The MICT® technology system is intended for use in the laboratory. It is an in vitro diagnostic device that provides quantitative and qualitative information regarding the presence of biochemical analytes that are labeled with a superparamagnetic particle (SPMP). The system accomplishes this by measuring analyte-specific MICT® Test Devices. The information provided by the MICT® technology system can be used as an aid in the diagnosis of specific disease or physiological states.
Electronically read lateral flow instruments typically employ optical transmission or reflectance techniques to quantify the accumulation of particles in the test region. Such techniques suffer from numerous inaccuracies which may be caused by:
The table below illustrates the differences between MICT® and conventional, particle-based immunoassays.
 | 1. Apply patient sample, wait for 10-20 minutes for test development. |
|---|---|
 | 2. Insert cassette into MICT® Instrument. |
 |
3. Press Start button to begin measurement. MICT® automatically recognizes the measurement parameters. |
 |
4. Read and Print results. |